Biocontrol and plant growth promoting potential of Trichoderma yunnanense
Karmakar Pintu, SenGupta Koyel, Das Panna, Bhattacharya Swati Gupta, Saha Ajay Krishna
Research Articles | Published: 05 July, 2021
First Page: 928
Last Page: 936
Views: 4525
Keywords: Trichoderma yunnanense , Internal transcribed spacer, Growth promoting fungi, Antagonism
Abstract
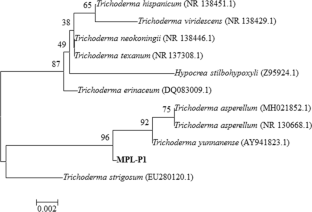
Biological control agents can be an effective and safe sustainable alternative method to reduce the detrimental effects of synthetic fungicides. In the present study, an airborne microfungi Trichoderma yunnanense showed broad spectrum antifungal and plant growth promoting activities. The characterization of the fungal isolate was confirmed by morphological features, DNA sequencing and phylogenetic tree analysis. Various growth parameters such as root and shoot length (13.76 mm and 19.3 mm), root and shoot dry weight (0.048 g and 0.037 g) were enhanced in the treated plants compare to control. In treated plants. The total chlorophyll and sugar content were found to be higher than control. Trichoderma yunnanense showed higher antagonistic potentiality (50–66%) against Alternaria brassicicola, Alternaria solani, Aspergillus ochraceus but showed lower potentiality against Pencillium oxalicum (21.42%). Thus the strain of Trichoderma yunnanense possessed growth-promoting potential and potent to antagonistic activity against plant pathogenic fungi.
References
Aban JL, Barcelo RC, Oda EE, Reyes GA, Balangcod TD, Gutierrez RM, Hipol RM (2017) Auxin production, phosphate solubilisation and ACC deaminase activity of root symbiotic fungi (RSF) from Drynaria quercifolia L. Bull Environ Pharmacol Life Sci 6:26–31
Adams P, de Leij FAAM, Lynch JM (2007) Trichoderma harzianum rifai 1295–22 mediates growth promotion of crack willow (Salix fragilis) saplings in both clean and metal-contaminated soil. Microb Ecol 54:306–313
Adhikari P, Oh Y, Panthee DR (2017) Current status of early blight resistance in tomato: an update. Int J Mol Sci 18(10):2019
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta Vulgaris. Plant Physiol 24:1–15
Badaluddin NA, Jamaluddin SN, Ihsam NS, Sajili MH, Mohamed NA (2018) Molecular identification of isolated fungi from Kelantan and Terengganu using internal transcribed spacer (ITS) region. J Agrobiotech 9:230–239
Bell DK, Wells HD, Markham CR (1982) In vitro antagonism of Trichoderma species against six fungal pathogens. Phytopathology 72:379–382
Contreras-Cornejo HA, López-Bucio JS, Méndez-Bravo A, MacíasRodríguez L, Ramos-Vega M, Guevara-García ÁA et al (2015) Mitogenactivated protein kinase 6 and ethylene and auxin signaling pathways are involved in Arabidopsis root-system architecture alterations by Trichoderma atroviride. Mol Plant-Microbe Interact 28:701–710
Erannna N, Mallikarjunaiah RR, Bagyaraj DJ, Suresh CK (2001) Response of Coleus aromaticus to Glomus fasciculatum and other beneficial soil microllora. J Spices Aromotic Crops 10:141–143
Farah ST, Nasreen S (2013) In vitro assessment of antagonistic activity of Trichoderma viride and Trichoderma harzianum against pathogenic fungi. Indian J App Res 3(5):57–59
Garnica-Vergara A, Barrera-Ortiz S, Muñoz-Parra E, Raya-González J, Méndez-Bravo A, Macías-Rodríguez L et al (2016) The volatile 6-pentyl-2H-pyran-2-one from Trichoderma atroviride regulates Arabidopsis thalianaroot morphogenesis via auxin signaling and ETHYLENE INSENSITIVE 2 functioning. New Phytol 209(4):1496–1512
González-Pérez E, Ortega-Amaro MA, Salazar-Badillo FB, Bautista E, Douterlungne D, Jiménez-Bremont JF (2018) The ArabidopsisTrichoderma interaction reveals that the fungal growth medium is an important factor in plant growth induction. Sci Rep 8:16427
Inbar J, Abramsky M, Cohen D, Chet I (1994) Plant growth enhancement and disease control by Trichoderma harzianum in vegetable seedlings grown under commercial conditions. Euro J Plant Path 100:337–346
Karmakar P, Debbarma P, Das Panna, Saha AK (2018) Isolation of soil fungi and screening of in vitro antagonistic potentiality of Trichoderma viride against Penicillium chrysogenum. Eur J Biomed Pharm Sci 5(2):883–888
Kimura M (1980) A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Köhl J et al (2010) Epidemiology of dark leaf spot caused by Alternaria brassicicola and A. brassicae in organic seed production of cauliflower. Plant Pathol 59:358–367
Konappa N, Krishnamurthy S, Dhamodaran N, Arakere UC, Chowdappa S, Ramachandrappa NS (2020) Opportunistic avirulent plant symbionts Trichoderma: exploring its potential against soilborne phytopathogens. In: Manoharachary C, Singh HB, Varma A (eds) Trichoderma: agricultural applications and beyond. Soil biology, vol 61. Springer, Cham
Murali M, Amruthesh KN, Sudisha J, Niranjana SR, Shetty HS (2012) Screening for plant growth promoting fungi and their ability for growth promotion and induction of resistance in pearl millet against downy mildew disease. J Phytol 4(5):30–36
Pandian RTP, Raja M, Kumar A, Sharma P (2016) Morphological and molecular characterizationof Trichoderma asperellum strain Ta13. Indian Phytopathol 69:297–303
Picos-Munoz PA, Garcia-Estrada RS, Carrillo-Fasio JA, Leon-Felix J, Allende-Molar R (2011) First report of blue mold caused by Penicillium oxalicum in Tomato (Solanum lycopersicum) in Mexico. Phytopathology 95:195.1-1195.1
Reddy BN, Saritha KV, Hindumathi A (2014) In vitro screening for antagonistic potential of seven species of Trichoderma against different plant pathogenic fungi. Res J Biol 2:29–36
Robinson GHJ, Balk J, Domoney C (2019) Improving pulse crops as a source of protein, starch and micronutrients. Nutr Bull 44(3):202–215
Roslee NR, Doni F, Auma K, Abzar A, Yusoff WMW (2017) Effect of Trichoderma spp. on mustard growth (Brassica juncea L.). Undergrad. Res J Biomol Sci Biotech 1:168–176
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular Cloning a laboratory manual. Cold Spring Harbor Laboratory Press, New York
Samuels GJ, Lieckfeldt E, Nirenberg HI (1999) Trichoderma asperellum, a new species with warted conidia, and redescription of Trichodermaviride. Sydowia 51:71–88
Smolińska U, Kowalska B (2018) Biological control of the soil-borne fungal pathogen Sclerotinia sclerotiorum a review. J Plant Pathol 100:1–12
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Taradolsirithitikul P, Sirisomboon P (2017) Qualitative and quantitative analysis of ochratoxin A contamination in green coffee beans using Fourier transform near infrared spectroscopy. J Sci Food Agric 97(4):1260–1266
Vincent JM (1947) Distortion of fungal hyphae in the presence of certain inhibitors. Nature 159:850–850
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR Protocols: a guide to methods and applications. Academic Press, San Diego, pp 315–322
Woo SL, Ruocco M, Vinale F, Nigro M, Marra R, Lombardi N et al (2014) Trichoderma-based products and their widespread use in agriculture. Open Mycol J 8:71–126
Yemn EW, Willis AJ (1954) The estimation of carbohydrate in plant extracts by/anthrone. Biochem J 57(3):508–514
Yu ZF, Qiao M, Zhang Y, Zhang KQ (2007) Two new species of Trichoderma from Yunnan, China. Antonie Van Leeuwenhoek 92:101–108
Zhang Y, Zhuang WY (2020) Trichoderma brevicrassum strain TC967 with capacities of diminishing cucumber disease caused by Rhizoctonia solani and promoting plant growth. Biol Control 142:104151
Author Information
Mycology and Plant Pathology Laboratory, Department of Botany, Tripura University, Agartala, India