Hierarchical clustering highlights root-specific biochemical responses to bean seed cryopreservation
*Article not assigned to an issue yet
Cejas Inaudis, Quintana-Zaez Julio César, Acosta Yanier, Companioni Barbarita, Zevallos–Bravo Byron E., de Lourdes Tapia y Figueroa María, Lorenzo José Carlos
Short Communications | Published: 14 April, 2026
First Page: 0
Last Page: 0
Views: 424
Keywords: Cryoconservation, Hierarchical cluster analysis, Liquid nitrogen exposure, Multivariate analysis, n Phaseolus vulgaris L
Abstract
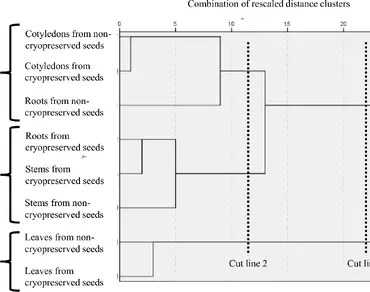
Hierarchical Cluster Analysis (HCA) is a technique for grouping objects based on their similarities, forming a hierarchical structure represented as a dendrogram. It follows two main strategies: agglomerative (bottom-up) and divisive (top-down). HCA is extensively applied across disciplines such as biology, marketing, and social sciences. In plant biotechnology, it plays a crucial role in assessing genetic diversity, phenotypic variations, metabolomic patterns, pathogen resistance, and environmental adaptability. Although HCA is not widely employed in plant germplasm cryopreservation research, it holds potential for evaluating the genetic diversity and stability of cryopreserved specimens. This communication illustrates the novel application of HCA in a common bean seed cryopreservation experiment. This study investigated the effects of cryopreservation on P. vulgaris seeds during early germination. No visible phenotypic changes were seen in seedlings from cryopreserved seeds. However, significant biochemical changes were noted: cryopreservation increased aldehydes (excluding malondialdehyde) in shoots from 56.47 to 253.19 µmol g− 1 fresh weight, and reduced phenolics in roots. Cut line 1 in the dendrogram identified two main groups, with leaves distinctly different from other organs. Cut line 2 separated non-cryopreserved roots in branch 1 and cryopreserved roots in branch 2. HCA, as a methodological tool, confirmed roots were more affected by cryostorage.
References
Acosta Y, Companioni B, Escalante D, Zevallos–Bravo BE, Pérez-Bonachea L, Chmielarz P, Hajari E, Neinhuis C, Melzer M, Lorenzo JC (2024) Scanning electron microscopy reveals contrasting effects of liquid nitrogen on seeds of legumes Neonotonia wightii, Phaseolus vulgaris and Tamarindus indica. Acta Physiol Plant 46:77. https://doi.org/10.1007/s11738-024-03703-2
Almeida J, Barbosa L, Pais A, Formosinho S (2007) Improving hierarchical cluster analysis: a new method with outlier detection and automatic clustering. Chemom Intell Lab Syst 87:208–217. https://doi.org/10.1016/j.chemolab.2007.01.005
Basak S, Ramesh AM, Kesari V, Parida A, Mitra S, Rangan L (2014) Genetic diversity and relationship of Hedychium from Northeast India as dissected using PCA analysis and hierarchical clustering. Meta Gene 2:459–468. https://doi.org/10.1016/j.mgene.2014.05.002
Białoskórska M, Rucińska A, Boczkowska M (2024) Molecular mechanisms underlying freezing tolerance in plants: implications for cryopreservation. Int J Mol Sci 25:10110. https://doi.org/10.3390/ijms251810110
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilising the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Cejas I, Vives K, Laudat T, González-Olmedo J, Engelmann F, Martínez-Montero ME, Lorenzo JC (2012) Effects of cryopreservation of Phaseolus vulgaris L. seeds on early stages of germination. Plant Cell Rep 31:2065–2073. https://doi.org/10.1007/s00299-012-1317-x
Chanana S, Thomas CS, Zhang F, Rajski SR, Bugni TS (2020) HCAPCA: automated hierarchical clustering and principal component analysis of large metabolomic datasets in R. Metabolites 10:297. https://doi.org/10.3390/metabo10070297
de Souza NMP, Dill RE, Fagundes DGdS, Cagliari A, Rieger A (2024) Multivariate analysis of physical–chemical and biological parameters along wheat growth (Triticum aestivum L.) and the effects of a phosphorus‐solubilizing bioinoculant. Ann Appl Biol 184:278–287. https://doi.org/10.1111/aab.12881
Deeb T, Brunotte R, Hubenia O, Leal-Marin S, Glasmacher B (2024) Advancements in cryogenic freezing instrumentation for cryopreservation. Ann Rev Heat Transf 27:2024053933. https://doi.org/10.1615/annualrevheattransfer.2024053933
Entensa Y, González-Morales A, Linares C, Vázquez JG, Martínez-Montero ME, Zevallos-Bravo BE, Hajari E, Vicente O, Villalobos-Olivera A, Lorenzo JC (2022) Exposure of Calophyllum antillanum seeds to liquid nitrogen delays seedling emergence and decreases the leaf levels of anthraquinones. CryoLetters 43:58–65. https://doi.org/10.54680/fr22110110812
Entensa Y, Lorente G, Pérez-Bonachea L, Rodolfo Ynchausti J, Martínez J, Companioni B, Hajari E, Acosta Y, Pritchard HW, Carlos Lorenzo J (2025) Exposure of carrot seeds to cryopreservation increases root weight and decreases levels of cell wall-linked phenolics. CryoLetters 46:135–142. https://doi.org/10.54680/fr25210110812
Figueiredo JRM, Paiva PDdO, da Silva DPC, Paiva R, Mesquita R, Souza RR, dos Reis MV (2021) Anatomical changes and cytogenetic stability in bird of paradise plants after zygotic embryo cryopreservation by desiccation method. In Vitro Cell Dev Biol Anim 57:272–280
Granato D, Santos JS, Escher GB, Ferreira BL, Maggio RM (2018) Use of principal component analysis (PCA) and hierarchical cluster analysis (HCA) for multivariate association between bioactive compounds and functional properties in foods: a critical perspective. Trends Food Sci Technol 72:83–90. https://doi.org/10.1016/j.tifs.2017.12.006
Gurr S, McPherson J, Bowles D (1992) Lignin and associated phenolic acids in cell walls. In: Wilkinson DL (ed) Molecular plant pathology. Oxford Press, Oxford, p 62
Heath R, Packer J (1968) Photoperoxidation in isolated chloroplast: I. kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Hong X, Wang J, Qi G (2014) Comparison of spectral clustering, K-clustering and hierarchical clustering on e-nose datasets: application to the recognition of material freshness, adulteration levels and pretreatment approaches for tomato juices. Chemom Intell Lab Syst 133:17–24. https://doi.org/10.1016/j.chemolab.2014.01.017
ISTA (2005) International rules for seed testing. International seed testing association, Bassersdorf, Switzerland
Jiang Y, Zhong G, Wang L, Wang T, Wang M, Zhang L, Zhou Y, Ding C, Yang R, Wang X (2014) The use of principal component analyses and hierarchical cluster analyses in the quality evaluation of Salvia miltiorrhiza Bunge. Qual Assur Saf Crops Foods 6:445–451. https://doi.org/10.3920/QAS2013.0275
Kantardzic M (2003) Data mining: concepts, models, methods and algorithms. Technometrics 45:277
Kaur J, Ghai N, Chahal GK, Sandhu S (2024) Characterization of maize inbred lines using principal component and cluster analysis for heat tolerance at the seedling stage under in vitro conditions. Cereal Res Commun 52:1431–1442. https://doi.org/10.1007/s42976-023-00468-1
Leal W, Llanos EJ, Restrepo G, Suárez CF, Patarroyo ME (2016) How frequently do clusters occur in hierarchical clustering analysis? A graph theoretical approach to studying ties in proximity. J Cheminform 8:1–16. https://doi.org/10.1186/s13321-016-0114-x
Liu C, Liu S, Tse WM, Tse KWG, Erbu A, Xiong H, Lanzi G, Liu Y, Ye B (2023) A distinction between Fritillaria cirrhosa bulbus and Fritillaria pallidiflora bulbus via LC–MS/MS in conjunction with principal component analysis and hierarchical cluster analysis. Sci Rep 13:2735. https://doi.org/10.1038/s41598-023-29631-8
Lorenzo JC, Acosta Y, Zevallos-Bravo BE, Chmielarz P, Hajari E, Höfer M, Ludwig-Mueller J (2023) Exposure of maize seeds to liquid nitrogen modifies the morphology and hormonal response of young plants. CryoLetters 44:369–377. https://doi.org/10.54680/fr23610110612
Nagel M, Pence V, Ballesteros D, Lambardi M, Popova E, Panis B (2024) Plant cryopreservation: principles, applications, and challenges of banking plant diversity at ultralow temperatures. Annu Rev Plant Biol 75:797–824. https://doi.org/10.1146/annurev-arplant-070623-103551
Ochatt S, Lambardi M, Panis B, Pathirana R, Revilla MA, Wang Q-C (2021) Cryopreservation and in vitro banking: a cool subject–preface from the editors. Plant Cell Tiss Org Cult 144:1–5. https://doi.org/10.1007/s11240-020-01985-1
Park J, Woo J, Choi G, Choi D, Jung M (2015) Chlorogenic acid profiles and antioxidant potentials of 17 sweet potato varieties cultivated in Korea: impact of extraction condition and classification by hierarchical clustering analysis. J Food Chem Nanotechnol 1:3–12. https://doi.org/10.17756/jfcn.2015-001
Porra R (2002) The chequered history of the development and use of simultaneous equations for the accurate determination of chlorophylls a and b. Photosynth Res 73:149–156. https://doi.org/10.1023/A:1020470224740
Ren R, Zhou H, Zhang L, Jiang X, Zhang M, Liu Y (2022) ROS-induced PCD affects the viability of seeds with different moisture content after cryopreservation. Plant Cell Tiss Org Cult 148:623–633. https://doi.org/10.1007/s11240-021-02219-8
Shan M-Q, Wang T-J, Jiang Y-L, Yu S, Yan H, Zhang L, Wu Q-N, Geng T, Huang W-Z, Wang Z-Z (2019) Comparative analysis of sixteen active compounds and antioxidant and anti-influenza properties of Gardenia jasminoides fruits at different times and application to the determination of the appropriate harvest period with hierarchical cluster analysis. J Ethnopharmacol 233:169–178. https://doi.org/10.1016/j.jep.2019.01.004
Stanwood P, Bass L (1981) Seed germplasm preservation using liquid nitrogen. Seed Sci Technol 9:423–437 (Corpus ID: 83112437)
Villalobos-Olivera A, González-Morales A, Martínez J, Gómez D, Martínez-Montero ME, Hajari E, Chmielarz P, Höfer M, Lorenzo JC (2023) The role of antioxidants, peroxidase and superoxide dismutase, in ameliorating oxidative stress associated with cryopreservation of Tamarindus indica seeds. Acta Physiol Plant 45:86. https://doi.org/10.1007/s11738-023-03569-w
Wang M-R, Bi W, Shukla MR, Ren L, Hamborg Z, Blystad D-R, Saxena PK, Wang Q-C (2021) Epigenetic and genetic integrity, metabolic stability, and field performance of cryopreserved plants. Plants 10:1889. https://doi.org/10.3390/plants10091889
Whelehan LM, Funnekotter B, Bunn E, Mancera RL (2022) The case for studying mitochondrial function during plant cryopreservation. Plant Sci 315:111134. https://doi.org/10.1016/j.cpb.2021.100019
Zhang Z, Murtagh F, Van Poucke S, Lin S, Lan P (2017) Hierarchical cluster analysis in clinical research with heterogeneous study population: highlighting its visualization with R. Ann Transl Med 5:75–81. https://doi.org/10.21037/atm.2017.02.05
Author Information
Department for Plant Breeding and Conservation of Genetic Resources, Bioplant Center, University of Ciego de Ávila Máximo Gómez Báez, Ciego de Avila, Cuba