Plant growth promoting traits of marine Micrococcus sp. with bio-control ability against Fusarium in chickpea plant
Patel Palak, Patel Keyur, Dhandhukia Pinakin, Thakker Janki N.
Research Articles | Published: 04 February, 2021
First Page: 94
Last Page: 101
Views: 4110
Keywords: Antifungal activity, Bio-control agent, IAA production, Marine organism, Plant growth promoting bacteria
Abstract
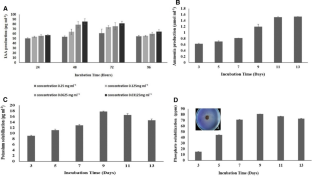
Chickpea is considered as one of the protein rich and low input crops in India but it suffers from a huge group of soil borne pathogens. Fusarium wilt, caused by Fusarium oxysporum f. sp. ciceris (FOC) at any time from the seedling to podding and become a major factor for yield loss. The aim of this study was to determine the plant growth promoting and bio-control activity of marine Micrococcus luteus and checking its in vitro effect on vegetative parameters in the presence and absence of pathogen. M. luteus was characterized as PGPB as it was tested positive for indole 3-acetic acid (IAA) production (81.379 µg ml−1 of IAA), ammonia production (1.513 µmol ml−1), potassium solubilization (17.78 µg ml−1), phosphate solubilization (81.03 ppm), glucanase production (0.5848 µmol ml−1 h−1) and chitinase production (0.024 µmol ml−1 h−1) along with antagonistic potential against four different soil borne pathogens. Talc-based bio-formulation was prepared using M. luteus and its effect on vegetative parameters was checked by performing pot trials, using four different conditions- control (T1), M. luteus treated (T2), FOC treated (T3) and both M. luteus and FOC treated (T4). M. luteus showed significantly positive effect on vegetative parameters of plants. The results suggested that M. luteus protected chickpea from pathogen as it showed bio-control ability with inhibitory effect against pathogens and act as PGPB to enhance overall plant growth.
References
- Agrawal T, Kotasthane AS (2012) Chitinolytic assay of indigenous trichoderma isolates collected from different geographical locations of Chhattisgarh in Central India. SpringerPlus 1(1):73. https://doi.org/10.1186/2193-1801-1-73
- Aleksandrov VG, Blagodyr RN, Ilev IP (1967) Liberation of phosphoric acid from apatite by silicate bacteris. Mikrobiolohichnyi Zhurnal (Kiev) 29:111–114
- Alexander M (1985) Introduction to soil microbiology. Wiley, USA, pp 382–385
- Aloni R, Aloni E, Langhans M, Ulrich CI (2006) Role of cytokinin and auxin in shaping root architecture: regulating vascular differentiation, lateral root initiation, root apical dominance and root gravitropism. Ann Bot 97:883–893. https://doi.org/10.1093/aob/mcl027
- Aneja KR (2009) Experiments in microbiology. New Age International Publications, New Delhi, India, pp 245–275
- Arrigo KR (2005) Marine microorganisms and global nutrient cycles. Nature 437(7057):349–355
- Brick JM, Bostock RM, Silverstone SE (1991) Rapid in situ assay for indole acetic acid production by bacteria immobilized on nitrocellulose membrane. Appl Environ Microbiol 57:535–538
- Cappucino JC, Sherman N (1992) Nitrogen cycle. Microbiology: a laboratory manual, 4th edn. Benjamin/Cumming, New York, pp 311–312
- Chhonkar PK, Subba-Rao NS (1967) Fungi associated with legume root nodules. Can J Microbiol 13(7):749–753. https://doi.org/10.1139/m67-099
- Datta J, Lal N (2012) Application of molecular markers for genetic discrimination of Fusarium wilt pathogen races affecting chickpea and pigeon pea in major region if India. Eur J Agron 58(1):55–65. https://doi.org/10.1170/T921
- Friedlander M, Inbar J, Chet I (1993) Biological control of soil borne plant pathogens by a β-1, 3 glucanase-producing Pseudomonas cepacia. Soil Biol Biochem 25(9):1211–1221. https://doi.org/10.1016/0038-0717(93)90217-Y
- Goswami D, Vaghela H, Parmar S, Dhandhukia P, Thakker JN (2013) Plant growth promoting potentials of Pseudomonas spp. strain OG isolated from marine water. J Plant Interact 8(4):281–290. https://doi.org/10.1080/17429145.2013.768360
- Graham PH, Vance CP (2000) Nitrogen fixation in perspective: an overview of research and extension needs. Field Crops Res 65:93–106. https://doi.org/10.1016/S0378-4290(99)00080-5
- John RP, Tyagi RD, Prévost D, Brar SK, Pouleur S, Surampalli RY (2010) Mycoparasitic Trichoderma viride as a biocontrol agent against Fusarium oxysporumf sp. adzuki and Pythium arrhenomanes and as a growth promoter of soybean. Crop Protect 29(12):1452–1459. https://doi.org/10.1016/j.cropo.2010.08.004
- King JE (1932) The colorimetric determination of phosphorus. Biochem J 26:292–297. https://doi.org/10.1042/bj0260292
- Kumar R, Mishra A, Jha B (2019) Bacterial community structure and functional diversity in subsurface seawater from the western coastal ecosystem of the Arabian Sea, India. Gene 701:55–64
- Marschner P (2012) Marschner’s mineral nutrition of higher plants, vol 3. Academic Press, London, pp 178–189
- Patel KB, Thakker JN (2019) Growth promotion and biocontrol activity of Nocardiopsis dassonvillei strain YM12: an isolate from coastal agricultural land of Khambhat. Vegetos 32(4):571–582
Author Information
Department of Biotechnology, P.D. Patel Institute of Applied Sciences, Charotar University of Science and Technology, Charusat Campus, Changa, India