Studies on development of pumpkin based antioxidant rich table wine
Sharma Somesh, Thakur Aman Deep, Chauhan Pankaj Kumar, Nagraik Rupak, Sharma Avinash, Kumar Dinesh, Thakur Nitika
Research Articles | Published: 27 July, 2020
First Page: 161
Last Page: 167
Views: 4576
Keywords: Pumpkin, Table wine, Herbs, Quality, Antioxidants, Phenols, Esters
Abstract
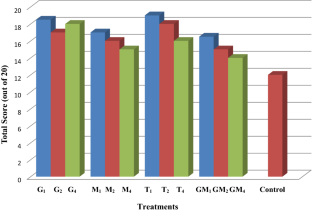
Pumpkin (Cucurbita moschata) is an important tropical vegetable found all around the world. In the present study, herbal enriched pumpkin wines were prepared using 1% pectinase, 0.50% acidulant and honey as sugar source along with the different concentration of dried herbs such as ginger (Zingiber officinale), basil leaves (Ocimum sanctum), licorice (Glycyrrhiza glabra) and their combinations. The herbal enriched pumpkin musts were fermented at a temperature of 28 °C. Further, on the basis of physicochemical and sensory characteristics, the treatment with Ocimum sanctum (basil leaves) 1% (T1) was adjudged as best. The wine had many desirable quality attributes such as a higher concentration of esters, phenols and low level of volatile acidity. The sensory scores also revealed that the herbal wine with basil leaves had high acceptability, with a moderate level of DPPH radical’s scavenging activity value. The wine prepared using Zingiber officinale and G. glabra (GM4) had the highest DPPH radical’s scavenging activity value (84%) and hence will scavenge more free radicals and is beneficial for health. It can be concluded that pumpkin as a base wine holds promise for the preparation of quality herbal enriched pumpkin table wine.
References
- AOAC (2004) Official methods of analysis. In: Hortwitz W (ed) Association of Official Analytical Chemists, 13th edn. North Fredrick Avenue Gaithersburg. https://doi.org/10.1002/0471740039.vec0284
- Amerine MA, Doughlas F (1974) Fermentation of grapes under anaerobic conditions. Holding grapes under CO2 before crushing. Am J Enol Vitic 25:1–6
- Amerine MA, Kunkee KE, Ough CS et al (1980) The technology of wine making, 4th edn. Publshing AVI. Co Inc, Westport, pp 794–798. https://doi.org/10.1002/food.19810251017
- Anonymous (2017) FAO Production year book, vol 52, Food and Agriculture organisation of the United Nations, Rome, pp 437–443. https://doi.org/10.1163/1570-6664_iyb_sim_org_2193
- Apeda (2015) Agri India Production Information from APEDA AgriExchange. https://apeda.gov.in/agriexchange/India/Production/
- Banerjee D, Chakrabarti S, Hazra AK et al (2008) Antioxidant activity and total phenolics of some mangroves in Sundarbans. Af J Biotechnol 7:805–810
- Blois MS (1958) Antioxidant determination by the use of stable free radicals. Nature 181:1199–2000. https://doi.org/10.1038/1811199a0
- Brand-Williams W, Cuvelier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. Leben WissUTech 28:25–30. https://doi.org/10.1016/s0023-6438(95)80008-5
- Busuricu F, Negranu-Parjol T, Balaban D et al (2008) The evaluation of the wines antioxidant activity of wine. J Agric Food Chem 43:1813–1819
- Caputi AJ, Ueda M, Brown J (1986) Spectrophotometric determination of ethanol in wine. Am J Enol Vitic 19:160–165
- Chopra RN, Chopra IC (1958) Indigenous Drugs of India, 2nd edn. Academic Publishers, Kolkata, pp183–187
- Deo B, Nath M, Nayak PK, Dhal Y (2013) Evaluation of antioxidant activity of Ocimum tenuiflorun, an important herb. Int J Plant Anim Environment Sci 3:150–154
- Guymon JF, Nakagiri J (1952) Methods for the determination of fusel oil. Proc Am J Enol Vitic 3:115–134
- Joshi VK, Sharma S, Bhushan S (2005) Effect of Method of Preparation and cultivar on the quality of Strawberry wine. Acta Aliment 34:339–353. https://doi.org/10.1556/aalim.34.2005.4.2
- Joshi VK (1997) Fruit Wines, 2nd edn, Directorate of Extn. Edu. Dr YS Parmar UHF, Nauni-Solan
- Joshi VK, Sharma S, Thakur A (2016) Wines–White, red, sparkling, fortified, and Cider. In: Pandey A, Du G, Sanroman MA, Soccol CR, Dussap CG (eds) Current developments in biotechnology and bioengineering (Elsevier book series of nine volumes). Elsevier, Amsterdam, pp 353–406. https://doi.org/10.1016/b978-0-444-63666-9.00013-3
- Joshi VK, Sharma S, Rana VS (2012) Wine and Brandy. In: Joshi VK, Singh RS (eds) Food biotechnology: principles and practices. IK International Publishing House, Pvt Ltd, New Delhi, pp 471–494. https://doi.org/10.1201/b12055-19
- Hopkins DL, Thomas CE (1996) Uses of Cucurbita moschata.. Am J Sci 56:328–332
- Liberaty V(1961) Ester determination and their applications to wine University of California, Davis M.Sc.,Thesis
- Mahant K, Sharma S, Sharma S, Thakur AD (2017) Effect of nitrogen source and citric acid addition on wine preparation from Japanese persimmon. J Inst Brew 123(1):144–150. https://doi.org/10.1002/jib.405
- Mahony OM (1985) Sensory evaluation of food: statistical methods and procedures, Ist edn. Marcel Dekker, Inc., New York, pp 168–169
- Maragatham C, Panneerselvam A (2011) Comparative analysis of papaya wine from other fruit wine. J Pur Appl Microbiol 5:967–969
- Markovic M, Bevanada Martiovic BA, Talics S (2015) Antioxidant activity and total phenol content of white wine Zilavka. Bull Chem Technol Bosnia Herzeg 44:1–4. https://doi.org/10.7868/s0015330315050127
- Maynard DN, Hochmuth GJ (2007) Knott’sHandbook for Vegetable Growers, 5th edn. John Wiley and Sons, Inc., Hoboken, pp 125–130. https://doi.org/10.1002/9780470121474
- Mota M, Besie JM, Srehaiano P, Goma G (1987) A simple device for fed-bach control in alcoholic fermentation. Biotechnol Bioeng 24:775–777. https://doi.org/10.1002/bit.260290619
- Ranganna S (1986) Handbook of analysis and quality control for fruit and vegetable products, 2nd edn. Tata McGraw Hill Publications. Co., New Delhi, pp 45–53
- Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphormolybadic phosphor tungstic acid reagents. Am J Enol Vitic 16:144–158. https://doi.org/10.5344/ajev.2016.16024e
- Sharma S, Thakur AD, Sharma S, Attenasova M (2018) Effect of yeast species on pumpkin wine making. J Inst Brew 124:187–193. https://doi.org/10.1002/jib.480
- Stoilova I, Krastanov A, Stoyanova A, Denev P, Gargova S (2007) Antioxidant activity of a ginger extract (Zingiber officinale). Food Chem 102: 764–770. https://doi.org/10.1016/j.foodchem.2006.06.023
- Thakur AD, Saklani A, Sharma S, Joshi VK (2014) Effect of different sugar sources, pectin esterase and acidulant concentration of pumpkin wine production. Int J Food Ferment Technol 4:67–78. https://doi.org/10.5958/2277-9396.2014.00007.5
- Thimmaiah SK (1999) Standard methods of biochemical analysis, 1st edn. Kalyani Publishers, New Delhi, pp 135–145
- Wills RBH (2007) Postharvest: An introduction to the physiology and handling of fruit and vegetables, 5th edn. AVI, UNSW Press, Boston, pp 543–581. https://doi.org/10.1079/9781845932275.0013
- Yildirim A, Mavi A, Kara A (2001) Determination of antioxidant and antimicrobial activities of Rumex crispus L. extracts. J Agric Food Chem 49:4083–4089. https://doi.org/10.1021/jf0103572
Author Information
Faculty of Applied Sciences and Biotechnology, Shoolini University, Solan, India