Therapeutic potential of crude extracts of Litchi chinensis seeds powder
Research Articles | Published: 25 March, 2024
First Page: 950
Last Page: 956
Views: 3224
Keywords: n L. chinensisn , Antioxidant, Polyphenols
Abstract
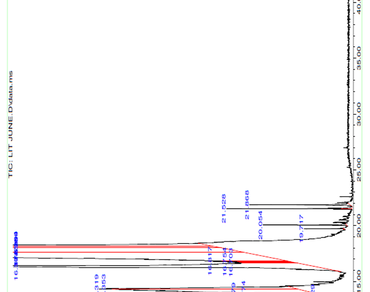
The purpose of the current study is to examine L. chinensis seed powder’s potential as a candidate for health promotion. Two distinct solvents viz. aqueous methanol and aqueous acetone were used to extract the sample. The nutritional parameters of the samples, such as their antioxidant capacity and the amount of phenolics, flavonoids, and flavonols, were examined. Thermal desorption coupled with GC/MSD allowed for the identification of oleo/aromatic compounds. Polyphenols were quantified using LCMS/MS. The findings indicated that the activities of the aqueous methanol extract were slightly superior. In 100 mg of methanol extract, the amounts of phenolic, flavonoid, and flavonol were found to be 72.99 mg (gallic acid), 27.1 mg (CE), and 11.45 mg (RE), respectively. The DPPH IC50 value for the antioxidant activity of methanol extract was found to be 0.049 mg ml− 1. Ascorbic acid, p-coumaric acid, gallic acid, ellagic acid, quercetin, and rutin were detected by LC-MS/MS analysis. According to the results of the GC-MS analysis, seed powder has good oleo/aromatic compounds and can be applied to a variety of cosmetic applications. The aforementioned findings demonstrate the high nutritional and therapeutic value of L. chinensis seed powder.
References
Ajila CM, Naidu KA, Bhat SG, Prasada R (2007) Bioactive compounds and antioxidant potential of mango peel extract. Food Chem 105:982–988. https://doi.org/10.1016/j.foodchem.2007.04.052
Almela L, Sanchez-Munoz B, Fernandez-Lopez JA, Roca M, Rabe V (2006) Liquid chromatographic mass spectrometric analysis of phenolics and free radical scavenging activity of rosemary extract from different raw materials. J Chromatogr 1120:221–229. https://doi.org/10.1016/j.chroma.2006.02.056Epub 2006 Mar 24
Azwanida NN (2015) A review on the extraction methods used in medicinal plants, principle, strength and limitation. Med Aromat Plants 4(3):1–6. https://doi.org/10.4172/2167-0412.1000196
Chang ST, Wu JH, Wang SY, Kang PL, Yang NS, Shyur LF (2001) Antioxidant activity of extracts from Acacia confusa bark and heartwood. J Agric Food Chem 49:3420–3424. https://doi.org/10.1021/jf0100907
Demiray S, Pintado ME, Castro LMP (2009) Evaluation of phenolic profiles and antioxidant activities of Turkish medicinal plants Tilia argentea, Crataegi folium leaves and Polygonum bistorta roots. World Acad Sci Eng Technol 54:312–317. https://doi.org/10.5281/zenodo.1075066
Dhan P, Garim U, Pushpangadana P (2011) Antioxidant potential of some under-utilized fruits. Indo-Global J Pharm Sci 1:25–32
Dorman HJD, Deans SG (2000) Antimicrobial agents from plants: antibacterial activity of plant volatile oils. J App Microbio 88. https://doi.org/10.1046/j.1365-2672.2000.00969.x. 308– 316
Gorinsteina S, Zachwiejab Z, Katricha E, Pawelzikc E, Haruenkitd R, Trakhtenberge S, Martin-Belloso O (2004) Comparison of the contents of the main antioxidant compounds and the antioxidant activity of white grapefruit and his new hybrid. J Food Sci Technol 37:337–343. https://doi.org/10.1016/j.lwt.2003.10.005
Guo H, Luo H, Yuan H, Xia Y, Shu P, Huang X, Lu Y, Liu X, Keller ET, Sun D (2017) Litchi seed extracts diminish prostate cancer progression via induction of apoptosis and attenuation of EMT through Akt/GSK-3 signaling. Sci Rep 7:41656. https://doi.org/10.1038/srep41656
Jurikova T, Skrovankova S, Mlcek J, Balla S, Snopek L (2019) Bioactive compounds, antioxidant activity, and Biological effects of European Cranberry (Vaccinium oxycoccos). Molecules 24(1):24. https://doi.org/10.3390/molecules24010024
Kilari E, Putta S (2016) Biological and phytopharmacological descriptions of Litchi chinensis. Phcog Rev 10:60. https://doi.org/10.4103/0973-7847.176548
Kunradi VFG, Da Silva Campelo Borges G, Copetti C, Da Valdemiro Gonzaga L, Costa Nunes E, Fett R (2009) Activity and contents of polyphenolic antioxidants in the whole fruit, flesh and peel of three apple cultivars. Arch Latinoam Nutr 59:101–106
Ma Q, Xie H, Li S, Zhang R, Zhang M, Wei X (2014) Flavonoids from the pericarps of Litchi chinensis. J Agric Food Chem 62:1073–1078. https://doi.org/10.1021/jf405750p
Miliauskas G, Venskutonis PR, Beek TAV (2004) Screening of radical scavenging activity of some medicinal and aromatic plant extract. Food Chem 85:231–237. https://doi.org/10.1016/j.foodchem.2003.05.007
Prasad K, Yang B, Yang S, Chen Y, Zhao M, Ashraf M, Jiang Y (2009) Identification of phenolic compounds and appraisal of antioxidant and antityrosinase activities from litchi (Litchi chinensis Sonn.) Seeds. Food Chem 116:1–7
Rangel JC, Benavides Jorge, Heredia JB, Zevallos LC, Luis, Velázquez DJ (2013) The Folin-Ciocalteu assay revisited: improvement of its specificity for total phenolic content determination. Anal Methods 5(21):5990. https://doi.org/10.1039/c3ay41125g
Rehman ZU (2006) Citrus peel extract–A natural source of antioxidant. Food Chem 99:450–454. https://doi.org/10.1016/j.foodchem.2005.07.054
Sharma A, Kumar RA, Sood S, Khulbe RK, Agrawal PK, Bhatt JC (2018) Evaluation of nutraceutical properties of finger millet genotypes from mid hills of Northwestern Himalayan region of India. Indian J Exp Biol 56(1):39–47
Sharma A, Rai PK (2018) Assessment of Bioactive compounds in Brassica juncea using chromatographic techniques. J Pharmacogn Phytochem 7(3):1274–1277
Shukla RK, Painuly D, Porval A, Shukla A (2012) Proximate analysis, nutritive value, total phenolic content and antioxidant activity of Litchi Chinensis Sonn. Nat Prod Indian J 8(9):361–369
Singh JP, Chandel R, Mishra B, Suneetha V (2013) Evaluation of antimicrobial and antioxidant property of lychee’s seed for therapeutic purpose. Int J Pharm Sci Rev Res 19:72–76
Author Information
Department of Bioscience & Biotechnology, Banasthali University, Jaipur, India