Standardization of adventitious root induction in stem cuttings of Cynometra travancorica Bedd Willd., an endangered tree species of Western Ghats
Hrideek T. K., Geethu P. D., Jijeesh C. M., Raghu A. V., Muraleekrishnan K.
Research Articles | Published: 08 February, 2019
First Page: 11
Last Page: 18
Views: 4152
Keywords: Cynometra travancorica , IBA, NAA, Propagation, Rooting
Abstract
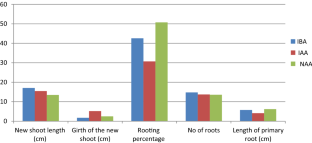
Cynometra travancorica Bedd. belonging to family Fabaceae is an endangered tree species endemic to Western Ghats with restricted distribution. There is an urgent need for conservation of this endangered species and the vegetative propagation techniques can play major role in planting stock production. Present study focuses on the standardisation of vegetative propagation protocol of C. travancorica using stem cuttings with different concentrations of three plant growth regulating substances in different season. The anatomical, physiological and biochemical changes of the sprouted cuttings belonging to different growth regulator treatments were also conducted. Softwood, semi hardwood and hardwood cuttings were collected in three seasons (January–April, May–August and September–December) and treated with indole 3 butyric acid, indole 3 acetic acid and naphthalene acetic acid in different concentrations. Rooting response was measured after 45 days in the mist chamber. The adventitious rhizogenesis of C. travancorica stem cuttings was influenced by type of cutting, season of collection, type of growth regulator and its concentration. Semi hardwood cuttings collected during season III treated with 8000 mg l−1 IBA recorded about 84% rooting. Anatomical studies on the sprouted cuttings treated with different growth regulator did not show any significant differences, whereas physiological and biochemical parameters of sprouted cuttings varied. Hence, we recommend this treatment combination for the large scale planting stock production.
References
- Arnon DI (1949) Copper enzymes in isolated chloroplasts and polyphenol oxidases in Beta vulgaris. Plant Physiol 24:1–5
- Barrs HD, Weatherly PE (1962) A re-examination of the relative turgidity techniques for estimating water deficits in leaves. Aust J Biol Sci 15:423–428
- Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–207
- Bunce JA (1984) Effect of humidity on photosynthesis. J Exp Bot 65:1245–1251
- Day JS, Loveys BR (1998) Effect of season of rooting on the ability of cuttings shrubs, Boronia megastigma and Hypocalymma angustifolium, Endl. (White myrtle). Aust J Exp Agric 38:201–206
- Desai MN, Chavan N (2010) Antibacterial activity and phytochemical screening of Cynometra iripa Kostel. Int J Pharma Biosci 1(3):1–4
- Desai MN, Chavan N (2011) Fatty acid profile of Cynometra iripa Kostel from seeds and leaves. Int J Pharma Res Dev 2(11):58–62
- Ebercon A, Blum A, Jordan WR (1977) A rapid colorimetric method for epicuticular wax content of Sorghum leaves. Crop Sci 17:179–180
- Hae M, Funnah SM (2011) The effect of propagation media and growth regulars on rooting potential of Kei apple (Dovyalis caffra) stem cuttings at different physiological ages. Life Sci J 8:91–99
- Hartmann HT, Kester DE, Davies FT, Geneve RL (2002) Plant propagation: principles and practices, 7th edn. Prentice Hall Inc, Upper Saddle River, pp 176–328
- Husen A, Pal M (2006) Variation in shoot anatomy and rooting behaviour of stem cutting in relation to age of donor plants in teak (Tectona grandis Linn. F). New For 31:57–73
- Johansen DA (1940) Plant microtechnique. Mc-Graw-Hill Book Company, New York
- Karaguzel O (1997) Studies on the propagation of bougainvilleas from cuttings. Ziraat Fak Derg Akeniz Univ 10:109–118
- Lowry OH, Rosewbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with Folin-phenol reagent. J Biol Chem 193:265–275
- Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31(3):426–428
- Naalamle AJ, Domonick JR, Bassuk N (2008) Adventitious root formation in stem cuttings of Quercus bicolor and Quercus macrocarpa and its relationship to stem anatomy. J Am Soc Hort Sci 133:479–486
- Pearse HL (1948) Growth substances and their practical importance in horticulture. Common Wealth Bureau of Horticulture and Pantation crops, East Malling, England
- Saumya MT, Surendran T, Sreena KR (2013) Vegetative propagation of Oroxylum indicum in different seasons. In: Proceedings 25th Kerala Science Congress, 29 Jan–1 Feb 2013, Thiruvananthapuram, pp 24–26
- Saumya MT, Jijeesh CM, Hrideek TK, Surendran T (2014) Standardization of propagation through cuttings in Salacia fruticosa Heyne ex Lawson: a medicinal plant endemic to Western Ghats. Int J Agric Environ Biotech 7(3):565–570
- Surendran T (1998) Vegetative propagation of asokam. Evergreen 41:9
- Weaver RJ (1972) Plant growth substances in Agriculture. WH Freeman and Company, San Francisco, pp 91–141
- Welch-Keesey M, Lerner BR (2002) New plants from cuttings. Purdue University Cooperative Extension Service, West Lafayette
- Zeng X, Lu QN (1988) Application of plant growth regulators in fruit trees. Agricultural Publishing House, Beijing
Author Information
Kerala Forest Research Institute, Thrissur, India