Transgenic lettuce (Lactuca sativa L.) harboring chitinase gene expressed resistance against a devastating fungus, Sclerotinia sclerotiorum
Research Articles | Published: 19 November, 2022
First Page: 1265
Last Page: 1274
Views: 4031
Keywords: Lettuce, Transformation, n Chitinase gene, Fungal resistance
Abstract
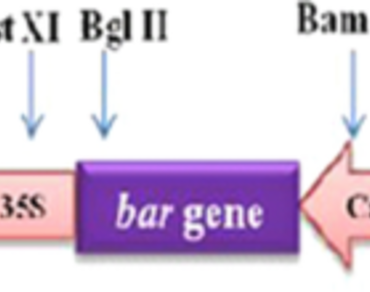
Lettuce (Lactuca sativa L.) is the world’s most popular leafy salad vegetable cultivated throughout the temperate region of the globe. However, its quality and yield are severely constrained by Sclerotinia sclerotiorum, a destructive fungal pathogen. In this study, rice chitinase (chi) gene was transferred to lettuce cv. Solan Kriti via Agrobacterium-mediated gene transfer technique to obtain resistance against S. sclerotiorum. To enhance the transformation rate, critical determinant variables including explant type, pre-culturing and co-cultivation time and acetosyringone effect were optimized. A combination of 72 h pre-culturing followed by 72 h co-cultivation period resulted in the highest transformation frequency of 8.60% and 3.31% in leaf and petiole explants, respectively. Furthermore, the transformation frequency was enhanced in leaf (23.22%) and petiole (16.44%) explants when the selection medium was fortified with acetosyringone in a concentration of 100 µM, respectively. Transgene (chi) integration was confirmed through PCR and Southern blotting. Expression of the chi gene in transgenic lettuce plants was confirmed by RT-PCR and qRT-PCR analysis. Fungal bioassay of transgenic lettuce proved the effectiveness of chi gene against invasion of Sclerotinia sclerotiorum by showing delay in symptom appearance. Lesions appeared within 12 h after incubation with the fungus in control plants, while leaves of transgenic plant formed lesion after 72 h, but no colonization of the foliar tissue was observed.
References
Ahmed MB, Akhter MS, Hossain M, Islam R, Choudhury TA, Hannan MM, Razvy MA, Ahmad I (2007) An efficient Agrobacterium-mediated genetic transformation method of lettuce (Lactuca sativa L.) with an aphidicidal gene, Pta (Pinellia ternata agglutinin). Middle-East J Sci Res 2(2):155–160
Chitrampalam P, Cox CA, Turini TA, Pryor BM (2010) Efficacy of Coniothrium minitans on lettuce drop caused by Sclerotinia minor in desert agro-ecosystem. Biol Control 55:92–96
Darqui FS, Radonic LM, Lopez N, Hopp HE, Bilbao ML (2018) Simplified methodology for large scale isolation of homozygous transgenic lines of lettuce. Electron J Biotechnol 31:1–9. https://doi.org/10.1016/j.ejbt.2017.10.002
Das MP, Rebecca LJ, Sharmila S, Banerjee A, Kumar D (2012) Identification and optimization of cultural conditions for chitinase production of Bacillus amyloliqufaciens SM3. J Chem Pharm Res 4:969–497
Dias BBA, Cunha WG, Morais LS, Vianna GR, Rech EL, de Capdeville GD, Aragao FJL (2006) Expression of an oxalate decarboxylase gene from Flammulina sp. in transgenic lettuce (Lactuca sativa) plants and resistance to Sclerotinia sclerotiorum. Plant Pathol 55:187–193
Fallah-Ziarani M, Haddad R, Garoosi G, Jalali M (2013) Agrobacterium-mediated transformation of cotyledonary leaf of lettuce (Lactuca sativa L.) by the GCHI gene. Iran J Genet Plant Breed 2(2):47–55
Gambhir G, Kumar P, Aggarwal G, Srivastava DK, Thakur AK (2020) Expression of cry1Aa gene in cabbage imparts resistance against diamondback moth (Plutella xylostella). Biol Future 71:165–173. https://doi.org/10.1007/s42977-020-00014-8
Gerszberg A (2018) Tissue culture and genetic transformation of cabbage (Brassica oleracea var. capitata): an overview. Planta 248:1037–1048
Girhepuje PV, Shinde GV (2011) Transgenic tomato plants expressing a wheat endochitinase gene demonstrate enhanced resistance to Fusarium oxysporum f. sp lycopersici. Plant Cell Tissue Organ Culture 105:243–251
Gomez KA, Gomez AA (1984) Statistical procedure for agricultural research. Willey, New York
Khan RS, Sjahril R, Nakamura I, Mii M (2008) Production of transgenic potato exhibiting enhanced resistance to fungal infections and herbicide applications. Plant Biotechnol Rep 2:13–20
Kreutz GF, Sandoya GV (2021) Exploring the potential of lettuce (Lactuca sativa L) as an early crop in Florida’s sandy soils. Hortic Sci 56(1):59–70. https://doi.org/10.21273/HORTSCI15420-20
Kumar P, Gambhir G, Gaur A, Sharma KC, Thakur AK, Srivastava DK (2018) Development of transgenic broccoli with cryIAa gene for resistance against diamondback moth (Plutella xylostella). 3 Biotech 8:299. https://doi.org/10.1007/s13205-018-1316-x
Mirzaee M, Jalali-Javaran M, Moieni A, Zeinali S, Behdani M (2018) Expression of VGRNb-PE immunotoxin in transplastomic lettuce (Lactuca sativa L.). Plant Mol Biol. https://doi.org/10.1007/s11103-018-0726-9
Mishra M, Jalil SU, Mishra RK, Kumari S, Pandey BK (2016) In vitro screening of guava plantlets transformed with endochitinase gene against Fusarium oxysporum f. sp. psidii. Czech J Genet Plant Breed 52:6–13
Mohebodini M, Jalali-Javaran M, Alizadeh H, Mahboudi F, Yarbakht M (2014) Agrobacterium-mediated transformation of lettuce (Lactuca sativa L.) to express IgG-binding protein A and human pro-insulin as a fusion protein. J Hortic Sci Biotechnol 89(6):719–725. https://doi.org/10.1080/14620316.2014.11513143
Mohebodini M, Javaran MJ, Mahboudi F, Alizadeh H (2011) Effect of genotype, explant age and growth regulators on callus induction and direct shoot regeneration of lettuce. Aust J Crop Sci 5:92–91
Odu NN, Okomuda MO (2013) Prevalance of Salmonella species and Escherichia coli in fresh cabbage and lettuce sold in Port Harcourt Metropolis, Nigeria. Rep Opin 5:1–8
Panara F, Calderini O, Porceddu A (2018) T-DNA insertional mutagenesis and activation tagging in Medicago truncatula. In: Cañas L, Beltrán J (eds) Functional genomics in Medicago truncatula. Methods in molecular biology, vol 1822. Humana Press, New York. https://doi.org/10.1007/978-1-4939-8633-0_6
Parmar N, Singh KH, Sharma D, Singh L, Kumar P, Nanjundan J, Khan YJ, Chauhan DK, Thakur AK (2017) Genetic engineering strategies for biotic and abiotic stress tolerance and quality enhancement in horticultural crops: a comprehensive review. 3 Biotech 7:239. https://doi.org/10.1007/s13205-017-0870-y
Pileggi M, Pereira AAM, Silva JS, Pileggi SAV, Verma DPS (2001) An improved method for transformation of lettuce by Agrobacterium tumefaciens with a gene that confers freezing resistance. Braz Arch Biol Technol 44:191–196
Sharma S, Srivastava DK (2017) Agrobacterium-mediated fungal resistance gene transfer studies pertaining to antibiotic sensitivity on cultured tissues of lettuce (Lactuca sativa L. cv. Solan Kriti). Int J Curr Microbiol App Sci 6(7):1687–1698
Sharma S, Kumar P, Gambhir G, Kumar R, Srivastava DK (2018) Assessment of genetic diversity in lettuce (Lactuca sativa L.) germplasm using RAPD markers. 3 Biotech 8(1):9. https://doi.org/10.1007/s13205-017-1039-4
Song D, Xiong X, Tu WF, Yao W, Liang HW, Chen FJ, He ZQ (2017) Transfer and expression of the rabbit defensin NP-1 gene in lettuce (Lactuca sativa). Genet Mol Res 16:333–337. https://doi.org/10.4238/gmr16019333
Srivastava DK, Kumar P, Sharma S, Gaur A, Gambhir G (2016) Genetic engineering for insect resistance in economically important vegetable crops. In: Ahmad N, Anis M (eds) Plant tissue culture: propagation, conservation and crop, vol 343. Springer, Singapore, p 378
Yang X, Yang J, Li H, Niu L, Xing G, Zhang Y, Xu W, Zhao Q, Li Q, Don Y (2020) Overexpression of the chitinase gene CmCH1 from Coniothyrium minitans renders enhanced resistance to Sclerotinia sclerotiorum in soybean. Transgenic Res 29:187–198. https://doi.org/10.1007/s11248-020-00190-2
Author Information
Department of Biotechnology, Dr. Y.S. Parmar University of Horticulture and Forestry, Solan, India