Establishing the dominating behavior of an aquatic plant ‘Najas marina’ L.
Research Articles | Published: 08 May, 2022
First Page: 1069
Last Page: 1077
Views: 3762
Keywords: Allelochemicals, Najas , Biodiversity, Hydrophytes, Weed, Wetland
Abstract
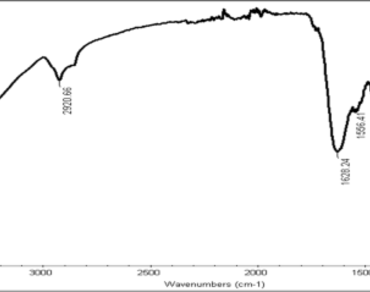
Through this study, an attempt has been made to understand the dominance of an obnoxious weed species Najas marina L., in Sukhna Lake, Chandigarh, India. The weed has been affecting the plant community in this water body. Phytochemical screening of the species in aqueous, ethanol and hexane solvents has revealed the presence of different phytoconstituents which may have their role in regulating its dominance. These chemicals are released into the water from the fragments or as leachates from decomposed plant parts. Gas chromatography–mass spectrometry (GC–MS) and liquid chromatography–mass spectrometry (LC–MS) were performed to get the detailed account of the chemical compounds present in this plant. Phenolics along with some specific chemical compounds, like Þ-hydroxybenzoic, Þ-coumaric, ferrulic, syringic acids etc. are to be responsible for the dominating behavior of N. marina. These phytoconstituents are possibly allelopathic in nature thus individually or in different combinations suppressing the growth of the adjoining species in the water body, and enable this species to flourish.
References
Agami M, Beer S, Waisel Y (1986) The morphology and physiology of turions in Najas marina L. in Israel. Aquat Bot 26:371–376
Ahluwalia AS (2012) Cyanobacterial and algal allelopathy. In: Cheema ZA et al. (eds) Allelopathy. Springer, New York, pp 1–23
Baratelli T, Gomes A, Wessjohann L, Kuster R, Simas N (2012) Phytochemical and allelopathic studies of Terminalia catappa L. (Combretaceae). Biochem Syst Ecol 41:119–125
Blum U, Gerig TM (2005) Relationships between phenolic acid concentrations, transpiration, water utilization, leaf area expansion and uptake of phenolic acids: nutrient culture studies. J Chem Ecol 31:1907–1932
Blum U, Gerig TM (2006) Interrelationships between þ-ccoumaric acid, evapotranspiration, soil water content and leaf expansion. J Chem Ecol 32:1817–1834
Caraco NF, Cole JJ (2002) Contrasting impacts of a native and alien macrophyte on dissolved oxygen in large river. Ecol Appl 12:1496–1509
Cipollini D, Stevenson R, Enright S (2008) Phenolic metabolites in leaves of the invasive shrub, Lonicera maackii and their potential phytotoxic and anti-herbivore effects. J Chem Ecol 34:144–152
Clifford MN, Wu WG, Kuhnert N (2006) The chlorogenic acids of Hemerocallis. Food Chem 95:574–578
Correll DL (1998) Role of phosphorus in the eutrophication of receiving waters: a review. J Environ Qual 27:261–266
Das B, Das R (1995) Chemical investigation in Parthenium hysterophorus L.—an allelopathic plant. Allelopathy J 2:99–104
Devmurari VP (2010) Phytochemical screening study and antibacterial evaluation of Symplocos racemosa Roxb. Arch Appl Sci Res 2:354–359
Dowd PF, Vega FE (1996) Enzymatic oxidation products of allelochemicals as a basis for resistance against insects: effects on the corn leaf hopper Dalbulus maidis. Nat Toxins 4:85–91
Dr. Duke’s Phytochemical and Ethnobotanical Databases (1992–2018) U. S. Department of Agriculture, Agricultural Science
El-Shahawy TA (2012) Allelopathic effect of certain local hydrophytes in Egypt for the potential uses in controlling weeds. J Appl Sci Res 8:4559–4567
Godghate A, Sawant R (2013) Qualitative phytochemical analysis of chloroform extract of leaves of Adhatodavasica Nees. Rasayan J Chem 6:107–110
Gross EM, Erhard D, Ivanyi E (2003) Allelopathic activity of Ceratophyllum demersum L. and Najas marina ssp. intermedia (wolfgang) Casper. Hydrobiologia 506:583–589
Harborne JB (1998) Phytochemical methods: a guide to modern techniques of plant analysis. Chapman & Hall Std., New York
Kanchan SD, Jayachandra (1979) Allelopathic effects of Parthenium hysterophorus L. III. Inhibitory effects of the weed residue. Plant Soil 53:37–47
Kaur S, Srivastava A, Kumar S, Srivastava V (2019) Biochemical and proteomic analysis reveals oxidative stress tolerance strategies of Scenedesmus abundans against allelochemicals released by Microcystis aeruginosa. Algal Res 41:1–11
Kpoviessi DSS, Accrombessi GC, Gbenou JD et al (2008) Cytotoxic activities of sterols and triterpens identified by GC-MS in Justicia anselliana (NEES) T. Anders active fractions and allelopathic effects on Cowpea (Vigna unguiculata (L.) Walp) plant. J Soc Ouest-Afr Chim 26:59–67
Kumar DG, Karthik M, Rajakumar R (2018) GC-MS analysis of bioactive compounds from ethanolic leaves extract of Eichhornia crassipes (Mart) Solms. and their pharmacological activities. Pharm Innov J 7:459–462
Kumbhar BA, Patel GR (2012) Effect of allelochemicals from Cressa cretica L. on in vitro pollen germination of Cajanus cajan (L.) Millsp. Bio Disc 3:169–171
Li FM, Hu HY (2005) Isolation and characterization of a noval antialgal allelochemical from Phragmites communis. Appl Environ Microbiol 71:6545–6553
Li ZH, Wang Q, Ruan X et al (2010) Phenolics and plant allelopathy. Molecules 15:8933–8952
Liu BY, Zhou PJ, Tian JR, Jiang SY (2007) Effect of pyrogallol on the growth and pigment content of cyanobacteria-blooming toxic and nontoxic Microcystis aeruginosa. Bull Environ Contam Toxicol 78:499–502
Mersie W, Singh M (1988) Effects of phenolic acids and Parthenium (Parthenium hysterophorus L.) extracts on tomato (Lycopersicon esculentum) growth and nutrient and chlorophyll content. Weed Sci 36:278–281
Mulderij G, Mau B, van Donk E, Gross EM (2007) Allelopathic activity of Stratiotesaloides on phytoplankton-towards identification of allelopathic substances. Hydrobiologia 584:89–100
Pandey DK (1994) Inhibition of Salvinia (Salvinia molesta Mitchell) by Parthenium (Parthenium hysterophorus L.). II. Relative effect of flower, leaf, stem and root residue on Salvinia and paddy. J Chem Ecol 20:3123–3131
Pandey DK (1996) Relative toxicity of allelochemicals to aquatic weeds. Allelopathy J 3:240–246
Pavia DL, Lampman GM, Kriz GS (2006) Introduction to Spectroscopy, 3rd edn. Thomson Business Information India Private Limited, India
Peng S, Huang J, Sheehy JE et al (2004) Rice yield decline with higher night temperature from global warming. Proc Natl Acad Sci USA 101:9971–9975
Prashanth N, Bhavani NL (2013) Phytochemical analysis of two high yielding Curcuma longa varieties from Andhra Pradesh. Int J Life Sci Biotechnol Pharm Res 2:103–108
Putnam AR (1988) Allelochemicals from plants as herbicides. Weed Technol 2:510–518
Putnam AR, Duke WB (1978) Allelopathy in agroecosystems. Annu Rev Phytopathol 16:431–451
Qiming X, Haidong C, Lijuan Q et al (2005) Allelopathic potential of aqueous extracts of submerged macrophytes against algal growth. Allelopathy J 15:95–104
Rice EL (1984) Allelopathy, 2nd edn. Academic Press, New York
Sakai T, Hayashi K (1973) Studies on the distribution of starchy and sugary leaves in monocotyledonous plants. Bot Mag 86:13–25
Seal AN, Pratley JE, Haig T, An M (2004) Identification of compounds in a series of allelopathic and non-allelopathic rice root exudates. J Chem Ecol 30:1647–1662
Shao-Lin P, Jun W, Qin-Feng G (2004) Mechanism and active variety of allelochemicals: a review. Acta Bot Sin 46:757–766
Sidhu MC, Puri S, Ahluwalia AS (2017a) Occurrence of invasive Najas marina (Hydrocharitaceae) in Sukhna Lake, Union Territory of Chandigarh, India. J Econ Taxon Bot 41:1–5
Sidhu MC, Puri S, Sharma A (2017b) Antibacterial activity, metabolites and elemental analysis of Saussurea candicans C. B. Clarke. Curr Bot 8:17–22
Silva SCA, Cervi AC, Bona C, Padial AA (2014) Aquatic macrophyte community varies in urban reservoirs with different degrees of eutrophication. Acta Limnol Bras 26:129–142
Strandas C, Kamal-Eldin A, Andersson R, Aman P (2008) Phenolic glucosides in bread containing flaxseed. Food Chem 110:997–999
Topuzovic MD, Radojevic ID, Dekic MS et al (2015) Phytomedical investigation of Najas minor All. in the view of the chemical constituents. EXCLI J 14:496–503
Ugochukwu SC, Uche IA, Ifeanyi O (2013) Preliminary phytochemical screening of different solvent extracts of stem bark and roots of Dennetia tripetala G. Baker. Asian J Plant Sci Res 3:10–13
Varjo E, Liikanen A, Salonen P, Martikainen PJ (2003) A new gypsum-based technique to reduce methane and phosphorus release from sediment of eutrophied lakes: (Gypsum treatment to reduce internal loading). Water Res 37:1–10
Wang H, Zhu H, Zhang K et al (2010) Chemical composition in aqueous extracts of Najas marine and Najas minor and their algae inhibition activity. In: Conference on environmental pollution and public health, pp 806–809
Weir L, Park S, Vivanco J (2004) Biochemical and physiological mechanisms mediated by allelochemicals. Curr Opin Plant Biol 7:472–479
Wium-Andersen S, Anthoni U, Houen G (1983) Elemental sulphur, a possible allelopathic compound from Ceratophyllum demersum. Phytochemistry 22:2613
Zhenbin W, Yunni G, Jing W, Biyun L, Qiahong Z (2009) Allelopathic effects of phenolic compounds present in submerged macrophytes on Microcystis aeruginosa. Allelopathy J 23:403–410
Zhu XF, Zhang HX, Lo R (2004) Phenolic compounds from the leaf extract of artichoke (Cynara scolymus L.) and their antimicrobial activities. J Agric Food Chem 52:7272–7278
Author Information
Department of Botany, Panjab University, Chandigarh, India