Phytochemical and pharmacological profiling from sporophylls of Isoetes coromandelina L. - leading to anti-inflammation studies
*Article not assigned to an issue yet
Research Articles | Published: 27 March, 2026
First Page: 0
Last Page: 0
Views: 101
Keywords: n Isoetes coromandelinan , Mineral analysis, Phytochemical profile, Antioxidant, Anti-inflammatory, Antimicrobial assay
Abstract
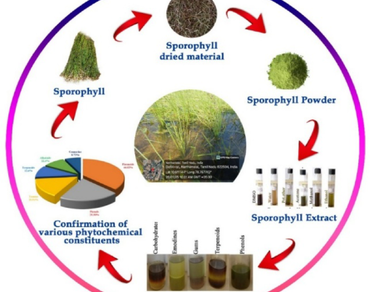
Isoetes coromandelina L., an aquatic Pteridophyte from the family Isoetaceae, has a history of traditional use as a remedy for rheumatism, tuberculosis and spleen and liver diseases. The present study is the first to investigate the phytochemical and pharmacological profiling from sporophylls of I. coromandelina, focusing on anti-inflammatory potential. Mineral analysis confirmed the presence of essential elements including Ca, K, NO3 and PO4. Phytochemical screening revealed diverse bioactive compounds such as flavonoids (46.02 mg), tannins (27.94 mg), phenols (28.30 mg), terpenoids (12.6 mg), alkaloids (11.6 mg), coumarins (10.72 mg), fatty acids, gums, catechins, emodin and anthocyanins. UV–Vis, FTIR confirmed major metabolites while HPLC specifically identified flavonoids and phenolic compounds. Pharmacological assessments demonstrated strong antioxidant activity through DPPH and hydroxyl radical scavenging assays, along with, significant anti-inflammatory activity using the HRBC membrane stabilization method with comparable IC50 values to standard references. The plant extract also exhibited notable antimicrobial activity against Staphylococcus aureus (8.3 ± 0.3 mm) and Candida albicans (11.6 ± 0.4 mm). These findings supports the use of I. coromandeina as a natural source as anti-inflammatory factor in tribal and ethnomedicine and hence could be used for further pharmaceutical and therapeutic applications in future. Explorations are needed to fulfil the lacuna in this area to tap the potential medicinal uses of this plant.
References
Aguilar EGP, Valmorida JS, Curayag QAL, Cruz RYD (2024) Quality evaluation of powdered fern (Diplazium esculentum): physicochemical, phytochemical, antioxidant, and anti-inflammatory properties in aqueous extracts. Mindanao J Sci Technol 22(S1):1–27
Ahmed MN, Gowan M, Azam MNK, Mannan M, Rahman M (2015) Clinical appraisals and phytochemical potential of ethnomedicinal pteridophyte: Drynaria quercifolia (L. (Polypodiaceae) Pharmacologyonline 1:4–17
Akinpelu LA, Olawuni IJ, Ogundepo GE, Adegoke AM, Olayiwola G, Idowu TO (2019) Spectroscopic analysis and anti-inflammatory effects of Milicia excelsa (Moraceae) leaf and fractions. GSC Biol Pharm Sci 6(3):051–060. https://doi.org/10.30574/gscbps.2019.6.3.0035
Al-khayri JM, Sahana GR, Nagella P, Joseph BV, Alessa FM, Al-mssallem MQ (2022) Flavonoids as potential anti-inflammatory molecules. Rev Molecules 27(9):2901. https://doi.org/10.3390/molecules27092901
Alcaráz LE, Blanco SE, Puig ON, Tomás F, Ferretti FH (2000) Antibacterial activity of flavonoids against methicillin-resistant Staphylococcus aureus strains. J Theor Biol 205(2):231–240. https://doi.org/10.1006/jtbi.2000.2062
Bahadori MB, Kordi FM, Ahmadi AA, Bahadori S, Valizadeh H (2015) Antibacterial evaluation and preliminary phytochemical screening of selected ferns from Iran. Res J Pharmacogn 2(2):53–59. http://rjpharmacognosy.ir
Bandyopadhyay A, Dey A (2022) Medicinal pteridophytes: ethnopharmacological, phytochemical, and clinical attributes. Beni-Suef Univ J Basic Appl Sci 11(1):113. https://doi.org/10.1186/s43088-022-00283-3
Baskaran XR, Geo Vigila AV, Zhang SZ, Feng SX, Liao WB (2018) A review of the use of pteridophytes for treating human ailments. J Zhejiang University-Science B 19(2):85–119. https://doi.org/10.1631/jzus.B1600344
Bhutia SP, Mohanty JP, Das R (2025) A comprehensive review on Diplazium esculentum : A traditional edible fern of North East India with broad pharmacological significance. J Med Plants Stud 13(4):14–19. https://doi.org/10.22271/plants.2025.v13.i4a.1885
Brinda P, Sasikala P, Purushothaman KK (1981) Pharmacognostic studies on Merugan kizhangu. Bull Med Eth Bot Res 3(1):84–96
Chen J, Yang J, Ma L, Li J, Shahzad N, Kim CK (2020) Structure-antioxidant activity relationship of methoxy, phenolic hydroxyl, and carboxylic acid groups of phenolic acids. Sci Rep 10(1):1–9. https://doi.org/10.1038/s41598-020-59451-z
Čtvrtlíková M, Kopáček J, Nedoma J, Znachor P, Hekera P, Vrba J (2023) Aquatic quillworts, Isoëtes echinospora and I. lacustris under acidic stress-A review from a temperate refuge. Ecol Evol. https://doi.org/10.1002/ece3.9878
Diniz RM, Gomes T, Fernandes F, Silva J, Mendonça P, Silva S, Felipe W, Saminez DS, Oliveira PVD, Alves E, Fonseca D, Santos C, Figueiredo S, Macêdo C, Filho B, Tereza M, Vanusa M, Cortez J, Sousa DS, Zagmignan A (2022) Antimicrobial and anti-infl ammatory effects of Eugenia brejoensis essential oil in mice wounds infected by Staphylococcus aureus. Front Pharmacol 13:999131. https://doi.org/10.3389/fphar.2022.999131
Dvorakova M, Soudek P, Pavicic A, Langhansova L (2024) The traditional utilization, biological activity and chemical composition of edible fern species. J Ethnopharmacol 324:117818. https://doi.org/10.1016/j.jep.2024.117818
Eltawaty SI, Omare MA, Almagboul AZ, Sanaa O (2019) Antimicrobial activity of leaves and bark of Libyan capparis spinosa subsp orientalis. Arab J Med Aromat Plants 4(2):42–56
Malavika G, Ravi SSS, Maheswary D, Leela KV, Harikumar Lathakumari R, Lekshmi PK (2025) Role of Candida albicans in chronic inflammation and the development of oral squamous cell carcinoma. Cancer Pathog Ther 3(5):402–410. https://doi.org/10.1016/j.cpt.2025.03.002
Garibo D, Borbón-Nuñez HA, de León JND, García Mendoza E, Estrada I, Toledano-Magaña Y, Tiznado H, Ovalle-Marroquin M, Soto-Ramos AG, Blanco A, Rodríguez JA, Romo OA, Chávez-Almazán LA, Susarrey-Arce A (2020) Green synthesis of silver nanoparticles using Lysiloma acapulcensis exhibit high-antimicrobial activity. Sci Rep 10(1):12805. https://doi.org/10.1038/s41598-020-69606-7
Halliwell B (2023) Understanding mechanisms of antioxidant action in health and disease. Nat Rev Mol Cell Biol 25(1):13–33. https://doi.org/10.1038/s41580-023-00645-4
Hilal B, Khan MM, Fariduddin Q (2024) Recent advancements in deciphering the therapeutic properties of plant secondary metabolites: phenolics, terpenes, and alkaloids. Plant Physiol Biochem 211:108674. https://doi.org/10.1016/j.plaphy.2024.108674
Huwae LMC, Silahooy VB (2024) Anti-inflammatory activity of fern Pteridium aquilinum ethanol extract gel on mice (Mus musculus) incision wounds. iop conf ser earth environ sci 1329(1):012002. https://doi.org/10.1088/1755-1315/1329/1/012002
Indumathi C, Durgadevi G, Nithyavani S, Gayathri PK (2014) Estimation of terpenoid content and its antimicrobial property in Enicostemma litorrale. Int J ChemTech Res 6(9):4264–4267
Iruthayamani SJ, Johnson M (2023) Uv-vis and ftir spectroscopic profile of gametophyte and sporophyte ethanolic extract of Anemia schimperiana c. presl subsp. wightiana (gardner) fraser-jenk. and cyathea gigantea (wall. ex. hook.) holtt. Rom J Biophys. https://doi.org/10.59277/RJB.2023.4.04
Khandelwal KR (2008) Practical Pharmacognosy. Pragati Books Pvt.Ltd
Kianifard T, Chopra A (2018) A therapeutic role for potassium (K) to reduce pain and complications related to the cardiovascular system and bone in rheumatoid arthritis (RA): A clinical research perspective. Rheumatol Res 3(1):1–12. https://doi.org/10.22631/rr.2017.69997.1035
Klein GL (2018) The role of calcium in inflammation-associated bone resorption. Biomolecules 8(3):4–9. https://doi.org/10.3390/biom8030069
Krishnaiah D, Devi T, Bono A, Sarbatly R (2009) Studies on phytochemical constituents of six Malaysian medicinal plants. J Med Plants Res 3(2):67–72
Liu Z, Hu L, Wang Q, Zhao X, Liu W, Zhang B, Pan W, Song W, Chen X, Zhang C, Wang J, Wang S, Zhou J (2025) Inorganic nitrate attenuates neuroinflammation after traumatic brain injury via Sialin. Tissue Cell 96:102955. https://doi.org/10.1016/j.tice.2025.102955
Makkar HPS (2003) Treatment of plant material, extraction of tannins, and an overview of tannin assays presented in the manual. Quantification Tannins Tree Shrub Foliage: Lab Man. https://doi.org/10.1007/978-94-017-0273-7_2
Manivannan V, Johnson M (2020) Total accepted phenolic, tannin, triterpenoid, flavonoid and sterol contents, anti-diabetic, anti-inflammatory and cytotoxic activities of Tectaria paradoxa (Fee.) sledge. Toxicol Rep 7:1465–1468. https://doi.org/10.1016/j.toxrep.2020.10.013
Manonmani R, Sara SC (2016) European journal of hplc finger prints of phenolic compounds present in. Actiniopteris radiata 3(6):435–438
Manonmani R, Sara SC (2013) Preliminary phytochemical analysis of Actiniopteris radiata (Swartz) Link. Int Res J Pharm 4(6):214–216. https://doi.org/10.7897/2230-8407.04648
Mendez-Encinas MA, Valencia D, Ortega-García J, Carvajal-Millan E, Díaz-Ríos JC, Mendez-Pfeiffer P, Soto-Bracamontes CM, Garibay-Escobar A, Alday E, Velazquez C (2023) Anti-inflammatory potential of seasonal sonoran propolis extracts and some of their main constituents. Molecules 28(11):4496. https://doi.org/10.3390/molecules28114496
Mohan PK, Krishna TPA, Thirumurugan A, Kumar TS, Kumari BDR Chemical profiling and in vitro antiurolithiatic activity of Pleurolobus gangeticus (L.) J. St.- Hil. ex, Ohashi H (2022) & K. Ohashi along with its antioxidant and antibacterial properties. Appl Biochem Biotechnol 194(11): 5037–5059. https://doi.org/10.1007/S12010-022-04017-0/Metrics
Nameni A, Arghavani S, Ghamari Kargar P, Beyzaei H, Ghani M, Maleki B (2025) HPLC analysis of phenolic compounds in Agaricus bisporus and Pleurotus ostreatus mushrooms: an assessment of their antibacterial capacities. Chem Methodol 9:343–355. https://doi.org/10.48309/chemm.2025.500231.1883
Osório ADC, Martins JLS (2004) Determinação de cumarina em extrato fluido e tintura de guaco por espectrofotometria derivada de primeira ordem. Rev Bras Ciências Farm 40(4):481–486. https://doi.org/10.1590/s1516-93322004000400005
Prasanna G, Anuradha R (2016) Ultraviolet - visible and fourier transform-infrared spectroscopic studies on Drynaria quercifolia L. rhizome. Methods 6(9):1–4
Pratiwi RA, Bayu A, Nandiyanto D (2022) How to read and interpret Uv-vis spectrophotometric results in determining the structure of chemical compounds. Indonesian J Educational Res Technol 2(1):1–20. https://doi.org/10.17509/ijert.v2i1.35171
Revathi MR, Catharin sara S (2012) Preliminary phytochemical analysis and antimicrobial activity of rhizome extract of Marsilea minuta, L. Int J Sci Res 3:46–48. https://doi.org/10.15373/22778179/feb2014/14
Sujatha S, Roselin R, Gayathri M, Sara S (2018) Preliminary phytochemical screening and spectroscopic analysis of Phymatosorus scolopendria (BURM. F.) Pic. Serm. Int Res J Pharm 9(7):168–172. https://doi.org/10.7897/2230-8407.097143
Schinella GR, Tournier HA, Prieto JM, Mordujovich De Buschiazzo P, Ríos JL (2002) Antioxidant activity of anti-inflammatory plant extracts. Life Sci 70(9):1023–1033.
Shirwaikar A, Devi S, Siju EN (2011) Anti-Inflammatory activity of Thespesia populnea fruits by membrane stabilization. Int J PharmTech Res 3(4):2060–2063.
Singh BP, Upadhyay R (2014) Medicinal Pteridophytes of Madhya Pradesh. J Med Plants Stud 2(4):65–68
Sundarajan P, Tamizhmani G, Srinivasan D, Rogers B (2016) Defects and statistical degradation analysis of photovoltaic power plants. Arizona State University
Singu BPSR, Upadhyay R (2013) 4.Ethnobotanical uses of Isoetes coromandelina L.F. and Actiniopteris radiata (Sw.) at Aravali hills Rampura region by Balendra Pratap Singh1_ Ranjana Singh2 and Ravi Upadhyay3. Life Sci Leafl 41:30–34
Van Buren JP, Robinson WB (1969) Formation of complexes between protein and tannic acid. J Agric Food Chem 17(4):772–777. https://doi.org/10.1021/JF60164A003/ASSET/JF60164A003.FP.PNG_V03
Wadaan MA, Baabbad A, Khan MF, Saravanan M, Anderson A (2023) Phytochemical profiling, anti-hyperglycemic, antifungal, and radicals scavenging potential of crude extracts of Athyrium asplenioides- an in-vitro approach. Environ Res 231:116129. https://doi.org/10.1016/j.envres.2023.116129
Waterman PG (1993) Phytochemical dictionary. A handbook of bioactive compounds from plants. Biochem Syst Ecol 21(8):849.
Author Information
PG and Research Department of Botany, Holy Cross College (Autonomous), Affiliated to Bharathidasan University, Tiruchirappalli, India