Aquaporin gene expression and antioxidant responses in sugarcane genotypes under water deficit during formative growth stage
*Article not assigned to an issue yet
Research Articles | Published: 31 March, 2026
First Page: 0
Last Page: 0
Views: 73
Keywords: Aquaporins, Water stress, Antioxidant activity, PIPs, Formative growth stage
Abstract
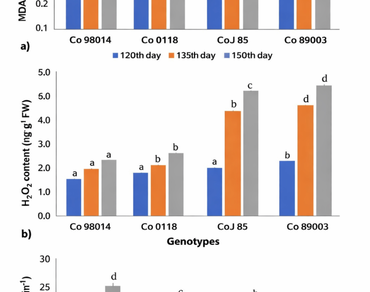
Water deficit in soil during the formative growth stage adversely affects the sugarcane productivity. Plants have developed a series of adaptive mechanisms to cope with these stresses. Characterization of plant genotypes under water deficit conditions will provide the basis for breeding new germplasms for efficient utilization of water and nutrients and adaptation to water stress. To achieve this goal, two tolerant (Co 98014 and Co 0118) and two sensitive (CoJ 85 and Co 89003) sugarcane genotypes, categorized in previous study were assessed for antioxidant responses, followed by differential expression of three aquaporin genes (ShPIP2-1, ShPIP-5 and ShPIP2-6) under two water deficit conditions. The MDA content was significantly increased 1.4 to 1.9 folds in tolerant genotypes than sensitive ones (0.4 to 0.7 folds). The percent change in H2O2 content was significantly increased upto 100.2 to 160.1% in the sensitive genotypes as compared to the tolerant genotypes (39.4 to 61.4%). The percent changes in SOD activity found greater (p < 0.05) in tolerant genotypes (136.9 to 233.5%) than the sensitive (66.3 to 137.2%). The transcript analysis of AQPs revealed upregulation of ShPIP2;5 but downregulation of ShPIP2;1 and ShPIP2;6 when plants were subjected to water deficit conditions. The findings suggested the role of antioxidant activity and PIP2 AQPs in mitigating oxidative stress under drought. This biological insight highlights that genotypes with stronger antioxidant defense and efficient aquaporin regulation are better equipped to maintain cellular homeostasis and water transport, thereby enhancing survival under water deficit. These traits can serve as valuable physiological markers for selecting drought‑tolerant sugarcane varieties, contributing to the development of resilient cultivars suited for semi‑arid regions.
References
Abbas S, Ahmad S, Sabir S, Shah A (2014) Detection of drought tolerant sugarcane genotypes (Saccharum officinarum) using lipid peroxidation, antioxidant activity, glycine-betaine and proline contents. J Soil Sci Plant Nutr 14:233–243
Ahmed F, Singh R, Kumar S (2025) Enhancing climate resilience in sugarcane: physiological and molecular responses to water stress. Front Plant Sci 16:145–160
Almeida RS, Oliveira RF, Silva MA (2013) Physio–biochemical responses of sugarcane under drought and salinity stress. Acta Physiol Plant 35:305–314
Andrade JCF, Terto J, Silva JV, Almeida C (2015) Expression profiles of sugarcane under drought conditions: variation in gene regulation. Genet Mol Biol 38(4):465–469. https://doi.org/10.1590/S1415-475738420140288
Bavai M, Ghasemi S, Khoshbakht K, Ghorbanpour M (2024) Investigation of biochemical and antioxidant system responses of sugarcane CP48-103 under drought stress induced by PEG. Int J Environ Sci Technol 22:3259–3270. https://doi.org/10.1007/s13762-024-05689-9
Beyer WF, Fridovich I (1987) Assaying for superoxide dismutase activity: some large consequences of minor changes in conditions. Anal Biochem 161:559–566
Boaretto LF, Carvalho G, Borgo L, Creste S, Landell MG, Mazzafera P, Azevedo RA (2014) Water stress reveals differential antioxidant responses of tolerant and nontolerant sugarcane genotypes. Plant Physiol Biochem 74:165–175
Chaves MM, Maroco JP, Pereira JS (2003) Understanding plant responses to drought-from genes to the whole plant. Funct Plant Biol 30:239–264
da Silva MD, de Andrade LM, de Souza CRB, de Oliveira JP (2013a) Expression analysis of sugarcane aquaporin genes under water deficit. J Nucleic Acids 2013:763293
de-Andrade LM, Nobile PM, Ribeiro RV, de-Oliveira JFNC, Figueira AVO, Frigel LTM, Nunes D, Perecin D, Brito MS, Pires RCM et al (2016) Characterization of PIP2 aquaporins in Saccharum hybrids. Plant Gene 31–37
dos-Santos CM, de-Almeida Silva M (2015) Physiological and biochemical responses of sugarcane to oxidative stress induced by water deficit and paraquat. Acta Physiol Plant 37:172
Galmes J, Pou A, Alsina MM, Tomas M, Medrano H, Flexas J (2007) Aquaporin expression in response to different water stress intensities and recovery in Richter-110: relationships with ecophysiological status. Planta 226:671–681
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Heinen RB, Ye Q, Chaumont F (2009) Role of aquaporins in leaf physiology. J Exp Bot 60:2971–2985
Huang GT, Ma SL, Bai LP (2012) Signal transduction during cold, salt, and drought stresses in plants. Mol Biol Rep 39(2):969–987
Kaldenhoff R, Fischer M (2006) Aquaporins in plants. Acta Physiol 187:169–176
Kumar D, Malik N, Sengar RS (2021a) Physio-biochemical insights into sugarcane genotypes under water stress. Biol Rhythm Res 52(1):92–115. https://doi.org/10.1080/09291016.2019.1587838
Kumar R, Singh RK, Singh P, Singh AK, Singh VP (2023) Drought and salinity stresses induced physio-biochemical changes in sugarcane: an overview of tolerance mechanism and mitigating approaches. Front Plant Sci 14:1225234. https://doi.org/10.3389/fpls.2023.1225234
Kumar D, Malik N, Sengar RS, Yadav B, Singh AK, Yadav CL, Yadav MK (2021b) Transcriptional regulation in sugarcane under water deficit during formative growth stage. Cytol Genet 55:598–605. https://doi.org/10.3103/S0095452721060062
Li GW, Zhang MH, Cai WM, Sun WN, Su WA (2008) Characterization of OsPIP2;7 a water channel protein in rice. Plant Cell Physiol 49:1851–1858
Lian LL, Yu X, Lane D, Sun WN, Tang ZC, Su WA (2006) Upland rice and lowland rice exhibited different PIP expression under water deficit and ABA treatment. Cell Res 16:651–660
Liu C, Liu Y, Guo K, Fan D, Li G, Zheng Y, Yu L, Yang R (2011) Effect of drought on pigments, osmotic adjustment and antioxidant enzymes in six woody plant species in karst habitats of Southwestern China. Env Exp Bot 71:174–183
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using realtime quantitative PCR and the 2ddCT methods. Methods 25:402–408
Locy RD, Hasegawa PM, Bressan RA (2002) Stress physiology. In: Taiz L, Zeiger E (eds) Plant Physiology, 3rd edn. Sinauer Associates, Inc. -Sunderland, pp 2438–2442
Mahendran G, Palanisamy S (2021) Aquaporin and its effect on foliar uptake to overcome drought stress in plants. Plant Physiol Rep 6(2):193–199. https://doi.org/10.1007/s40502-021-00592-0
Manimekalai R, Narayanan J, Gokul M, Selvi A, Gomati R, Kumar RA (2018) Biochemical and physiological response to oxidative stress in cultivated sugarcane and wild genera. Ind J Plant Physiol 23(2):261–270
Maurel C, Verdoucq L, Luu D, Santoni V (2008) Plant aquaporins: membrane channels with multiple integrated functions. Annual Rev Plant Biol 59:595–624
Miller G, Suzuki N, Ciftci-Yilmaz S, Mittler R (2010) Reactive oxygen species homeostasis and signaling during drought and salinity stresses. Plant Cell Environ 33:453–467
Pagariya MC, Harikrishnan M, Kulkarni PA, Devarumath RM, Kawar PG (2011) Physio-biochemical analysis and transcript profiling of Saccharum officinarum L. submitted to salt stress. Acta Physiol Plant 33(4):1411–1424
Patade VP, Bhargava S, Suprasanna P (2011) Salt and drought tolerance of sugarcane under iso-osmotic salt and water stress: growth, osmolytes accumulation, and antioxidant defense. J Plant Interact 6:275–282
Rodrigues FA, de-Laia ML, Zingaretti SM (2009) Analysis of gene expression profiles under water stress in tolerant and sensitive sugarcane plants. Plant Sci 176:286–302
Sakurai J, Ishikawa F, Yamaguchi T, Uemura M, Maeshima M (2005) Identification of 33 rice aquaporin genes and analysis of their expression and function. Plant Cell Physiol 46:1568–1577
Siddiqui MH, Al-Khaishany MY, Al-Qutami MA, Al-Whaibi MH, Grover A, Ali HM, Al-Wahibi MS, Bukhari NA (2015) Response of different genotypes of faba bean plant to drought stress. Int J Mol Sci 16:10214–10227
Silva MD, Silva RLO, Ferreira-Neto JRC, Guimarães ACR, Veiga DT, Chabregas SM, Burnquist WL, Kahl G, Benko-Iseppon AM, Kido EA (2013b) Expression analysis of sugarcane aquaporin genes under water deficit. J Nucleic Acid. https://doi.org/10.1155/2013/763945
Sultan M, Hui L, Yang LJ, Xian ZH (2012) Assessment of drought tolerance of some Triticum L. species through physiological indices. Czech J Genet Plant Breed 48(4):178–184
Veljovic-Jovanovic S, Noctor G, Foyer CH (2002) The potential influence of artifactual interference by tissue phenolics and ascorbate. Plant Physiol Biochem 40:501–507
Yepes-Molina L, Pérez-Pérez ME, García-Sánchez MJ, Olmos E (2020) Controversial regulation of gene expression and protein transduction of aquaporins under drought and salinity stress. Plants 9(12):1662. https://doi.org/10.3390/plants9121662
Author Information
Department of Agricultural Biotechnology, College of Agriculture, Sardar Vallabhbhai Patel University of Agriculture & Technology, Meerut, India